Our technology enables the controlled release of your asset by combining biological safety with precise structural engineering
Built on a protein with an established regulatory profile, ALBUPAD offers Pharma partners a safe, scalable, and differentiating platform for next-generation therapeutics.
Albumin is inherently biocompatible, non-immunogenic, and widely used in approved formulations. Yet its potential as a structured solid matrix has remained inaccessible due to the lack of a controllable shaping technology. ALBUPAD fills this innovation gap by engineering albumin into stable solid forms – microdiscs, films, and 3D matrices – with tunable mechanical properties, porosity, and degradation kinetics.
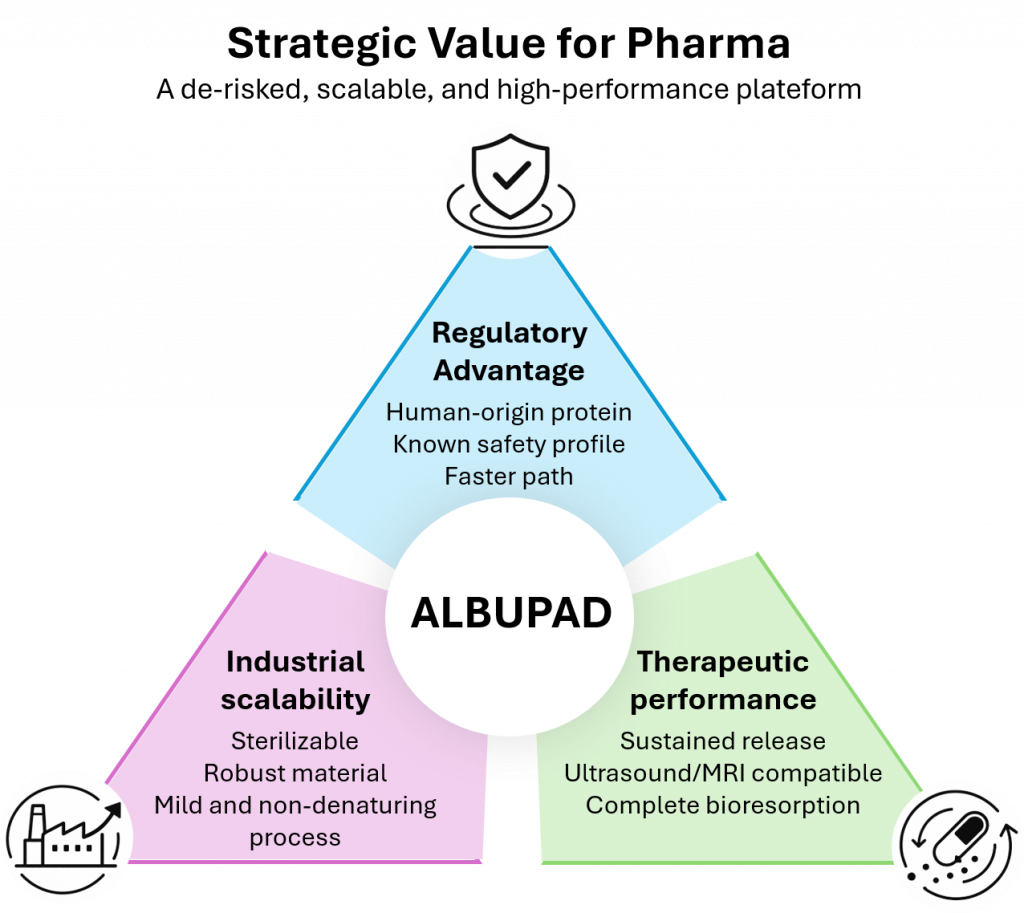
ALBUPAD provides key strategic advantages to Pharma companies:
- a clean regulatory path thanks to a human-origin excipient already familiar to health authorities;
- a scalable process to manufacture a biomaterial that withstands standard sterilization process, without loss of integrity or functionality, and that is inherently visible by ultrasound or MRI
- a proprietary programmable material capable of supporting sustained release, targeted delivery – which may benefit from the spontaneous targeting of cancer tissues by Albumin – and combination-device formats;
- a versatile platform that can be rapidly adapted to multiple therapeutic areas, types of active principles – such as small molecules, peptides, proteins or nucleotides, and routes of administration.
Why partner with us?
We bring a validated and data-rich platform, supported by integrated in vitro and in vivo evidence. Our in-house prototyping capabilities ensure agility and full coverage of preclinical development needs. Through strong academic and industrial partnerships, we access a broad ecosystem of equipment and expertise. Our proof of concept confirms the versatility of our technology to efficiently encapsulate and deliver diverse therapeutic compounds.
Our collaboration pathway
Initial engagement → NDA execution → Feasibility study (technical validation of encapsulation, release kinetics, and prototype development) → Joint development (process development, scale-up, and regulatory alignment toward GLP/GMP) → Licensing and technology transfer
Feasibility Validation
TRL 1 – 4 (3 à 6 months)
Co-Development
TRL 5 GLP (12 à 24 months)
Out Licencing
TRL 6 – 8 GLP / GMP
CORE PATENT
APPLICATION SPECIFIC PATENTS
